
The cycle is concerned with the formation of an ionic compound from the reaction of a metal (often a Group I or Group II element) with a halogen or other non-metallic element such as oxygen.īorn–Haber cycles are used primarily as a means of calculating lattice energy (or more precisely enthalpy ), which cannot otherwise be measured directly. It was also independently formulated by Kasimir Fajans and published concurrently in the same issue of the same journal. It was named after two German scientists, Max Born and Fritz Haber, who developed it in 1919. Charge polarisation allows there to be a degree of sharing of electrons, representing some covalent bonding, rather than pure ionic bonding.The Born–Haber cycle is an approach to analyze reaction energies. 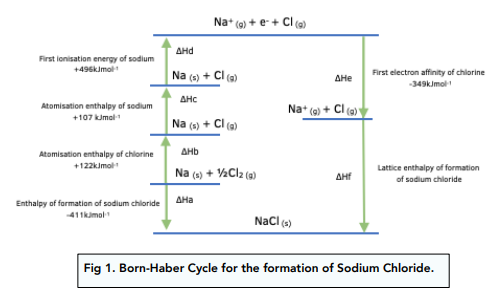
This phenomenon approaches electron sharing.Larger, positively charged ions are more polarisable. Chlorine ions are large and hence are easily polarised. Magnesium ions can closely approach the electron density of the negative chloride ions and polarise their charge. Smaller, positively charged ions are more polarising. This is due to magnesium ions being relatively small with a highly positive charge of +2. This is evidence that there are stronger bonding forces present than purely ionic. This is evidence of covalent character in the bonding within ionic lattices.Įxample: Magnesium Chloride Thermodynamic – Calculations involving Born-Haber Cyclesįor MgCl2, the experimental lattice enthalpy of formation is more exothermic. The experimentally calculated value is more exothermic than the theoretical value.This is evidence that many ionic compounds in fact show some degree of covalent character. The perfect ionic model is entirely theoretical, and it is seen that there is deviation from experimentally calculated values of lattice enthalpy using a Born-Haber cycle: The experimentally calculated value is more exothermic than the theoretical value. 
There is no covalent character in the bonds, it is purely ionic.The attractions are purely electrostatic.All ions have an even charge distribution.This is because many ionic compounds are not purely ionic but show some covalent character. Lattice enthalpies from Born-Haber cycles are sometimes higher than the values calculated from assumptions based on the perfect ionic model. Therefore ΔfH = -787kJmol-1 so the lattice enthalpy of sodium chloride is -787 kJ mol-1.Ĭomparing Lattice Enthalpies from Born-Haber Cycles 
To work out the value of ΔHf we are going to use the other values in the cycle. ΔHf represents the lattice enthalpy of formation of sodium chloride. Thermodynamic – Calculations involving Born-Haber Cycles Let’s revisit the Born-Haber Cycle for NaCl we looked at in the previous Chapter. If you have the values for all of the stages but one, you can work out the value of the unknown stage, using the principle of Hess’s Law. This process is made up of different stages, each of which corresponds to a different enthalpy change. Hess’s Law states that the total enthalpy change of a chemical reaction is the same independent of the route taken from products to reactants.īorn-Haber cycles illustrate the relationships between the different enthalpy changes involved in the formation of an ionic compound. This is possible due to the principle of Hess’s Law. Thermodynamic - Calculations involving Born-Haber Cycles (A-Level Chemistry) Calculations involving Born-Haber Cycles Calculations with Born-Haber Cycles Key TermsĪ Born-Haber cycle is a thermochemical cycle that includes every enthalpy change that is involved in an ionic compound formation process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
